- Visibility 68 Views
- Downloads 18 Downloads
- DOI 10.18231/j.ijpo.2024.050
-
CrossMark
- Citation
Clear cell adenocarcinoma with hepatoid differentiation of gallbladder: An unique histopathological type: Case with review of literature
Introduction
Clear cell gallbladder carcinoma was first reported in 1926 by Tyson and Piney and described a unique type of gallbladder tumor as hypernephroma of the gallbladder.[1] Clear cell variant of adenocarcinoma with hepatoid differentiation accounts for less than 1% of all gallbladder malignancies.[2] It is a very rare, distinctive type of gallbladder and extrahepatic bile duct malignant tumor. In the literature <18 cases have been reported. These are having an aggressive biologic behavior, delayed diagnosis at advanced stage therefore shows poor prognosis. The rising incidence and high mortality rate of biliary tract cancers demands urgency to search for early diagnosis.
Case Report
A 56-year old male patient presented with chief complaints of pain in the right upper quadrant of abdomen, vomiting since 15 days. Pain was moderate in nature and increasing in intensity. Associated with history of anorexia, weight loss and passing of dark yellow urine and yellowish discoloration of the sclera. Patient was known case of diabetes mellitus and was on regular treatment.
Patient’s liver function test showed serum albumin of 3.0 g/dl, serum alkaline phosphatase 1019 u/L, serum direct bilirubin3.1 mg/dl, serum aspartate aminotransferase 174 IU/L, serum alanine transaminase 146 IU/L, serum indirect bilirubin 2.4 mg/dl, serum total bilirubin of 5.5 mg/dl, serum total proteins of 6.6 g/dl, serum globulins 3.6 g/dl. Serum Alpha-fetoprotein level was elevated (480 ng/mL) the fasting blood sugar level was 270 mg/dl and HbA1C level was 13.7%. Hepatitis B surface antigen and ant hepatitis C antibody test were negative.
On ultrasonography abdomen- pelvis were suggestive of gall bladder neoplasm with cholelithiasis. The computer tomography-CT, MRCP abdomen-pelvis showed a distended gall bladder, with hyper tense circumferential wall thickening and a polypoidal and intramural growth seen in gall bladder fundus measuring 2.8x1.1x1.0 cm. Lesion was extending in adjacent segment IV B and V of liver. Few enlarged, pericholecystic, periportal and portocaval lymph nodes were noted. Reported as carcinoma of the gall bladder with liver extension and regional lymph node involvement. There was no distant metastases. The rest of pelvic organs were normal.
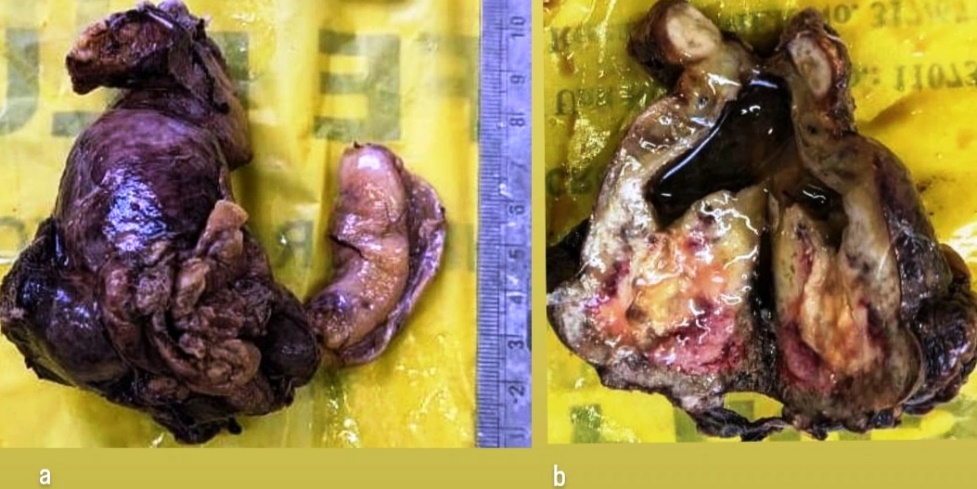
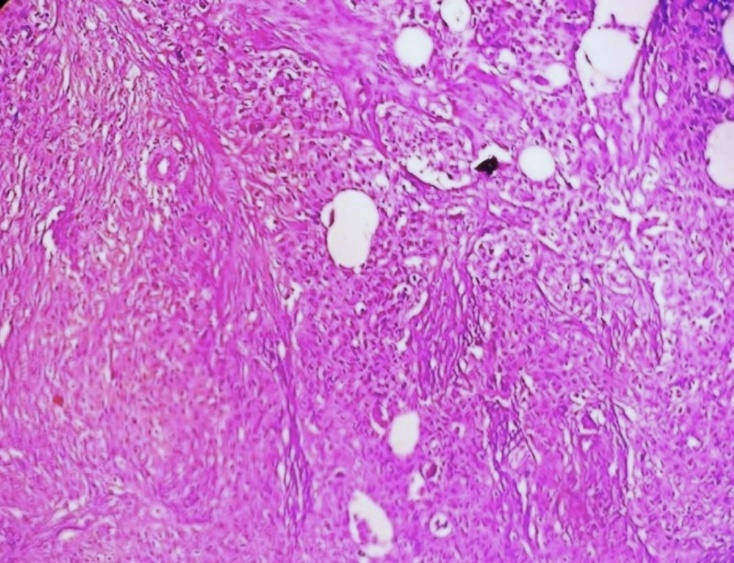
Patient underwent an open cholecystectomy with lymph nodes resection. We received, on gross the gall bladder, measured 8.2× 5.2×3 cm. On cutting open, showed a single, large, irregular, grey white to tan white, friable, polypoidal tumor measuring 3.5 x 2 x 1.5 cm, at body and fundus of gall bladder. Lumen was filled with yellow mucous. The tumor was extension to gall bladder wall with areas of hemorrhage, necrosis ([Figure 1] a, b). The surrounding irregular mucosal thickening and ulceration was noted. On microscopy showed a tumor composed of neoplastic cells arranged in an alveolar pattern, solid nests and trabeculae separated by blood vessels ([Figure 2] a, b). The >60% tumor showed clear cells with abundant clear cytoplasm and well-defined cytoplasmic boundaries and mucin vacuoles occasionally. The cytological atypia, large hyper chromatic nuclei with occasional nucleoli were noted. A few mitoses were noted. In areas foci of conventional adenocarcinoma of intestinal type with focal mucin production was observed. The tumor with areas of hepatoid differentiation were noted. The tumor was diffusely infiltrating the gallbladder wall ([Figure 3]). The hepatic tissue in adjacent IV B and V segment of liver and metastases in regional lymph nodes.

Discussion
Gallbladder tumors are the fifth most common cancers of the gastrointestinal tract. Usually these are detected incidentally, up to 50% in routine cholecystectomy specimens or in late stages. They have poor prognosis and low survival rate. Most patients diagnosed in the sixth or seventh decade of life with female preponderance.[3]
The various risk factors for the disease include cholelithiasis primary sclerosing cholangitis, polyps, choledochal cysts, typhoid, genetic etc.[4] The location of tumor with in the gallbladder is at the fundus (60%), body (30%) or neck (10%). Advanced tumors involve the liver and extra hepatic biliary tree. In our case tumor was invading the hepatic tissue in adjacent IV B and V segment of liver.
The various gross appearances in gallbladder carcinoma are polypoidal, infiltrating, and diffuse thickness of wall with variable ulcerations.
The histopathological type, adenocarcinoma is the most common, 98% of all gallbladder malignancy. The histologic variants of adenocarcinoma are pancreatobiliary, mucinous, clear cell carcinoma, intestinal, Hepatoid, sarcomatoid, and signet ring cell type.[5], [6] The uncommon types includes carcinosarcoma, small cell carcinoma, signet ring cell type, neuroendocrine tumours, lymphoma etc.
The adenocarcinomas are of well, moderate and poorly differentiated types. Clear cell adenocarcinoma of the gallbladder is an exceedingly rare malignant neoplasm that has been recognized by the World Health Organization as a distinctive type of gallbladder and extra hepatic bile duct tumor.[7] It is observed that the clear-cell variant is often associated with areas of classical adenocarcinoma, adenosquamous carcinoma, or mucinous carcinoma.
Bittinger et al. suggested the term clear cell adenocarcinoma to only those tumors manifesting clear cell differentiation in >50% of tumor cells.[8] In our case 60% tumor cells showed clear cell differentiation. In our case the tumor containing areas of hepatoid differentiation were noted. H C. Zhang et al reported a case of clear cell adenocarcinoma of the gallbladder with hepatoid differentiation, and mentioned that the clinical features of clear cell adenocarcinoma with hepatoid differentiation remain unclear due to the extremely rare incidence.[9]
Vardaman C et al. examined 550 cases of gallbladder carcinoma in which one case was reported with hepatoid differentiation.[10] In our case the associated adenocarcinoma was moderately differentiated. The stroma showed desmoplasia. On immunohistochemical stain tumor shows strong and diffuse CK7 and p53 positivity.
The differential diagnosis for clear-cell adenocarcinoma of the gallbladder are metastasis from renal-cell carcinoma, clear-cell carcinoma of the ovary, endometrial clear-cell carcinoma, clear-cell variant of hepatocellular carcinoma, malignant clear-cell sugar tumor of the lung.[11], [12]
Our case was conventional non-papillary gallbladder adenocarcinoma, which considered to have aggressive dissemination potential. The tumor of poor differentiation, having vascular invasion, and advanced stage have a higher chance of recurrence and have poorer outcomes. Various studies for the expression of prognostic biomarkers HER2/neu, KRAS and VEGF-A may be used in gallbladder cancer.[13] For hepatoid differentiation immunohistochemically shows tumor cell positivity for Hep Par1, AFP, alpha-1-antitrypsin, albumin, transferrin, PIVKA and, have been detected in the tumor cell cytoplasm.[14] The overall 5 year survival rate is of < 10% in gallbladder carcinoma. The clear cell adenocarcinoma variant of gallbladder carcinoma is exceedingly rare with less known about its recurrence and overall survival. The clear cell adenocarcinoma of gallbladder with hepatoid differentiation is an aggressive tumor and carries a bad prognosis.[15]
Conclusion
We are presenting this review of moderately differentiated clear cell adenocarcinoma of gallbladder with hepatoid differentiation having aggressive biologic behavior for its rarity, clinical, radiological, histopathological findings.
Conflict of Interest
None.
Source of Funding
None.
References
- W Tyson, A Piney. Hypernephroma of the gallbladder: cholecystectomy. Br J Surg 1926. [Google Scholar]
- R Maharaj, C Cave, K Sarran, N Bascombe, D Dan, W Greaves. A case report of the clear cell variant of gallbladder carcinoma. Int J Surg Case Rep 2017. [Google Scholar]
- MK Bind, RR Mishra, V Kumar, V Misra, PA Singh. Serum CA 19-9 and CA 125 as a diagnostic marker in carcinoma of gallbladder. Indian J Pathol Microbiol 2021. [Google Scholar]
- R Hundal, EA Shaffer. Gallbladder cancer: epidemiology and outcome. Clin Epidemiol 2014. [Google Scholar]
- SV Jagtap, JS Tele, SR Rajput, SS Jagtap. Mucinous Adenocarcinoma of Gall Bladder- A Rare Histopathological Variant Presented as Perforation. J Clin Diagn Res 2019. [Google Scholar]
- RD Goldin, JC Roa. Gallbladder cancer: a morphological and molecular update. Histopathology 2009. [Google Scholar]
- J Albores-Saavedra, DE Henson, LH Sobin. The WHO histological classification of tumors of the gallbladder and extrahepatic bile ducts. A commentary on the second edition. Cancer 1992. [Google Scholar]
- A Bittinger, I Altekrüger, P Barth. Clear cell carcinoma of the gallbladder: a histological and immunohistochemical study. Pathol Res Pract 1996. [Google Scholar]
- C Zhang, W Zhang, D Mu, X Shi, L Zhao. A clear cell adenocarcinoma of the gallbladder with hepatoid differentiation: case report and review of literature. Onco Targets Ther 2016. [Google Scholar]
- C Vardaman, S J Albores. Clear cell carcinomas of the gallbladder and extrahepatic bile ducts. Am J Surg Pathol 1995. [Google Scholar]
- P Warhate, SV Jagtap, S Kanetkar. Clinicopathological study of renal tumours in surgically excised specimens. Int J Res Pharm Sci 2020. [Google Scholar]
- M Ghaouti, K Znati, A Jahid, F Zouaidia, Z Bernoussi, Y El Fakir. A gallbladder tumor revealing metastatic clear cell renal carcinoma: report of case and review of literature. Diagn Pathol 2013. [Google Scholar]
- P Singh, SL Jain, P Sakhuja, A Agarwal. Expression of VEGF-A, HER2/neu, and KRAS in gall bladder carcinoma and their correlation with clinico-pathological parameters. Indian J Pathol Microbiol 2021. [Google Scholar]
- LM Terracciano, K Glatz, P Mhawech, M Vasei, FS Lehmann, R Vecchione. Hepatoid adenocarcinoma with liver metastasis mimicking hepatocellular carcinoma: an immunohistochemical and molecular study of eight cases. Am J Surg Pathol 2003. [Google Scholar]
- JS Su, YT Chen, RC Wang, CY Wu, SW Lee, TY Lee. Clinicopathological characteristics in the differential diagnosis of hepatoid adenocarcinoma: a literature review. World J Gastroenterol 2013. [Google Scholar]
